Overview of Chemicals and Products


A battery separator is a polymeric microporous foil that is positioned between the anode and the cathode. This positioning helps prevent electrical short-circuiting.
See the separator animation movie below or on YouTube
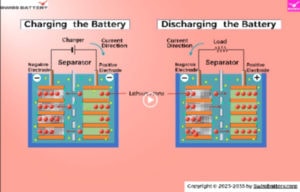
The porous battery separators allow ions of the electrolyte to pass through the battery separator. The battery separator is ionically conductive and electrically an isolator.
Read more extensively about Battery–Separators >>
Battery Separators are:
Read more about Battery-Separators here >>
A battery membrane is an often polymeric or composite foil that is positioned between the anode and the cathode. This positioning helps prevent electrical short-circuiting.
A battery membrane is a selective barrier-foil – it allows some ions to pass through but stops others.
Compared to battery separators, membranes are much more complex in their morphological and chemical structure. However, they are regularly non-porous.
Swiss Battery has developed an Ultrathin ion-selective polymer-membrane for high-power application.
Download the scientific article here.

Battery Membranes are:

Battery-Electrolyte are usually solution inside a battery cell. Depending on the type of battery or accumulator, it is being a liquid or paste-like (Gel-like) substance. The liquid consists of a solvent and a salt which is the source for Cations and anions.
Yet, no matter the type of battery or accumulator, the electrolyte serves the same purpose: it allows all ions to move between the cathode and the anode
Moreover, the electrolytes help to charge-compensate the active materials. Ions are electrically charged molecules or atoms.
These electrolyte-salts are solved in liquids (solvents, used in Lithium-Ion and Lead-acid), gelled and in dry formats. Electrolytes also can be polymers, ceramics (Solid-State-Battery) and molten salts (Sodium-Sulfur battery) .
Battery Electrolytes are:
The current collector collects the electrons (“the current”) from the electrode materials (anode and cathode) and passes them to the external circuit.
The current collectors are foils made of a highly electrically and often thermally conductive material, often a metal like copper and aluminum.

The current collectors are responsible to transport all current in the battery cell. The “active battery materials” are orders of magnitude less conductive and could not operate without the current collectors.

Polyelectrolytes are polymers with repeating units that bear an electrolyte group. These electrolyte groups are of an anionic or cathonic nature. Polyelectrolytes therefore have properties of both electrolytes and polymers, and have a high molecular weight.
Poly-electrolytes are often divided into three types, depending on the primary purpose of their application. These units are:
1. Polycation. For example, Polypyridinium salts and Polybenzimidazole (PBI) salts are bearing cathionic groups and are polycations.
2. Polyanions. For example, poly(sodium styrene sulfonate) (PSS) oder polyanion, poly acrylic acid (PAA) and und DNA-single strands oder DNS-double strands.
3. Ampholytic polymers. A polymer containing both cationic and anionic groups, or corresponding ionizable group.
Depending on the chemical structure of the ionic groups, the ampholytic polymers are called i) zwitter-ionic polymer, ii) polymeric inner salt, or iii) poly-betaine.
Sometimes Polymer-Electrolyte are referring to a mixture of a neutral Polymer and electrolyte salt. Polyethylenoxide (PEO) and Lithium-salts is a typical “polymer-electrolyte composite” used in rechargeable polymer-lithium ion batteries.
Polyelectrolytes are
Battery-Solvents are the liquid substance inside a battery cell. Depending on the type of battery or accumulator, it is an organic liquidor it is water. The solvent is responsible for the high conductivity of the dissolved electrolyte salt.

Classes of organic solvents to make liquid batter electrolyte:
1. Carbonates
2. Ether
Battery-Solvents are also used for manufacturing the battery electrodes cathode and anode. The solvent is used to form a a homogenous conductive electrode paste which is coated onto currents collectors.

Battery and Accumulator- Solvents are:


Lithium-ion batteries and other electronic devices are technically advanced and their functionally is complex. Higher Ampere ranges (current) and signal traffic and higher power and processing speeds have led to an increase in heat generation by batteries and electronic devices.

Thermal management has become of high importance,.
Therefore, the demand for thermal management using thermally conductive resins, composites, fillers, and glue is of high importance.
Anodes…

Anodes that are currently used are:

Cathodes are….

Battery Cathodes are:

Lithium-metal is used…

Applications for Lithium-metal are:

Ionic Liquids are promising…

Ionic liquids are:
BMS, short for Battery Management System…
Battery Management System…

Solid-sate-electrolytes are often polymeric or composites hat is positioned between the anode and the cathode. This positioning helps prevent electrical short-circuiting.
Solid-state electrolyte:
Conductive Carbon Materials…
Conductive Carbon Materials are:
A battery separator is a polymeric microporous foil that is positioned between the anode and the cathode. This positioning helps prevent electrical short-circuiting.
The porous battery separators allow ions of the electrolyte to pass through the battery separator. The battery separator is ionically conductive and electrically an isolator.
Battery Separators are:

A battery membrane is an often polymeric or composite foil that is positioned between the anode and the cathode. This positioning helps prevent electrical short-circuiting.
A battery membrane is a selective barrier-foil – it allows some ions to pass through but stops others.
Compared to battery separators, membranes are much more complex in their morphological and chemical structure. However, they are regularly non-porous.
Battery Membranes are:
The current collector collects the electrons (“the current”) from the electrode materials (anode and cathode) and passes them to the external circuit.
The current collectors are foils made of a highly electrically and often thermally conductive material, often a metal like copper and aluminum.
The current collectors are responsible to transport all current in the battery cell. The “active battery materials” are orders of magnitude less conductive and could not operate without the current collectors.
Battery-Electrolytes serve as a charge-transporting media to make a battery conductive by promoting the movement of ions from the cathode to the anode on charge and in reverse on discharge.
The electrolytes help to charge-compensate the active materials. Ions are electrically charged molecules or atoms which form salt at room temperature.
These electrolyte-salts are solved in liquids (solvents, used in Lithium-Ion and Lead-acid), gelled and in dry formats. Electrolytes also can be polymers, ceramics (Solid-State-Battery) and molten salts (Sodium-Sulfur battery) .
Battery Electrolytes are:
Redox-Flow Batteries (RFBs) are…
Battery Separators are:

Conducting Polymers is an often polymeric or composite foil that is positioned between the anode and the cathode. This positioning helps prevent electrical short-circuiting.
A battery membrane is a selective barrier-foil – it allows some ions to pass through but stops others.
Compared to battery separators, membranes are much more complex in their morphological and chemical structure. However, they are regularly non-porous.
Battery Membranes are:
Battery-Electrolytes serve as a charge-transporting media to make a battery conductive by promoting the movement of ions from the cathode to the anode on charge and in reverse on discharge.
The electrolytes help to charge-compensate the active materials. Ions are electrically charged molecules or atoms which form salt at room temperature.
These electrolyte-salts are solved in liquids (solvents, used in Lithium-Ion and Lead-acid), gelled and in dry formats. Electrolytes also can be polymers, ceramics (Solid-State-Battery) and molten salts (Sodium-Sulfur battery) .
Battery Electrolytes are:
The current collector collects the electrons (“the current”) from the electrode materials (anode and cathode) and passes them to the external circuit.
The current collectors are foils made of a highly electrically and often thermally conductive material, often a metal like copper and aluminum.
The current collectors are responsible to transport all current in the battery cell. The “active battery materials” are orders of magnitude less conductive and could not operate without the current collectors.



We help business to become sustainable